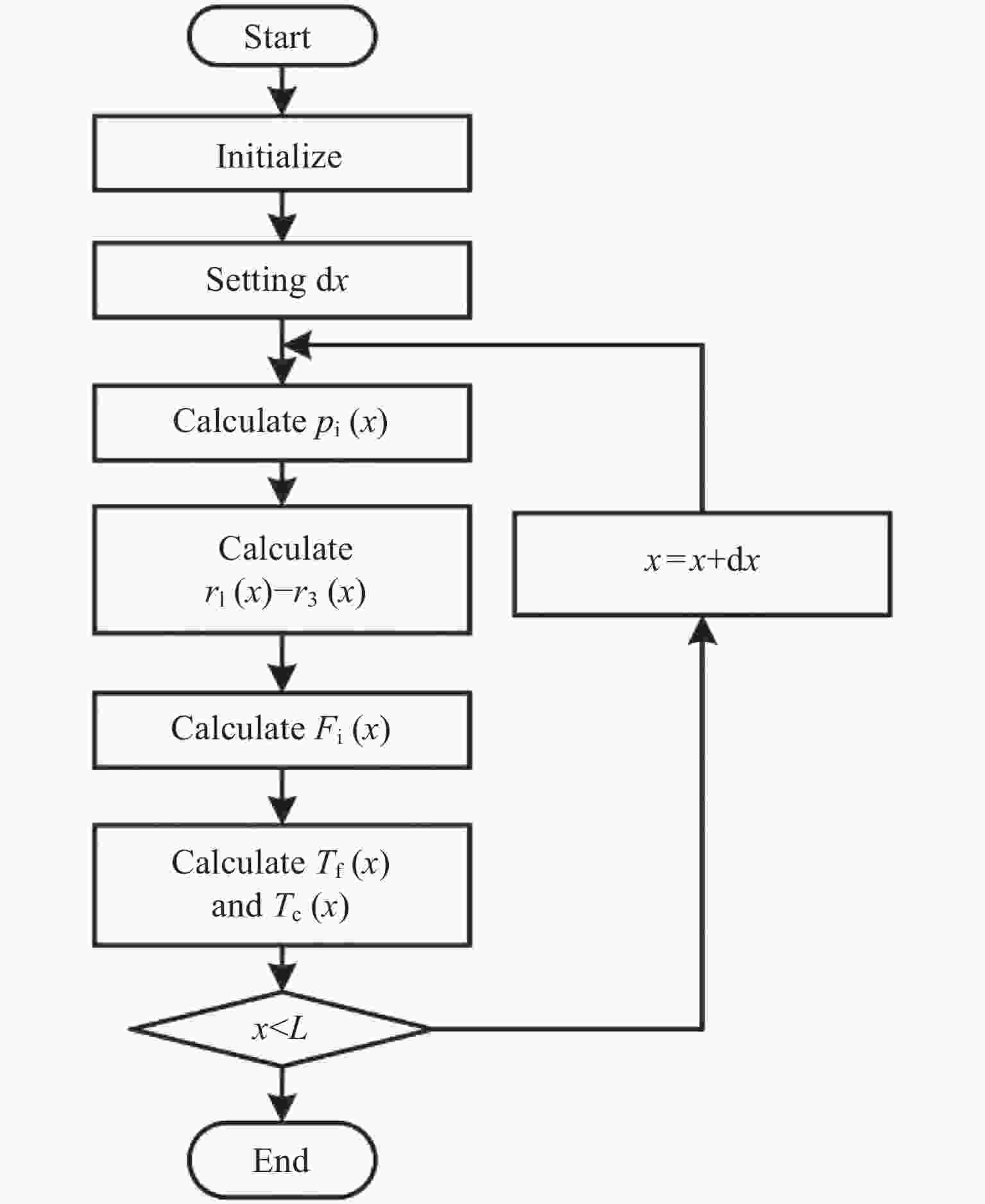
密闭空间内氢气和二氧化碳甲醇化系统的非均相模型及反应特性
doi: 10.11728/cjss2022.03.210123036 cstr: 32142.14.cjss2022.03.210123036
Numerical Study on Reaction Characteristics of Carbon Dioxide Hydrogenation to Methanol in Habitable Confined Space Based on Heterogeneous Models
-
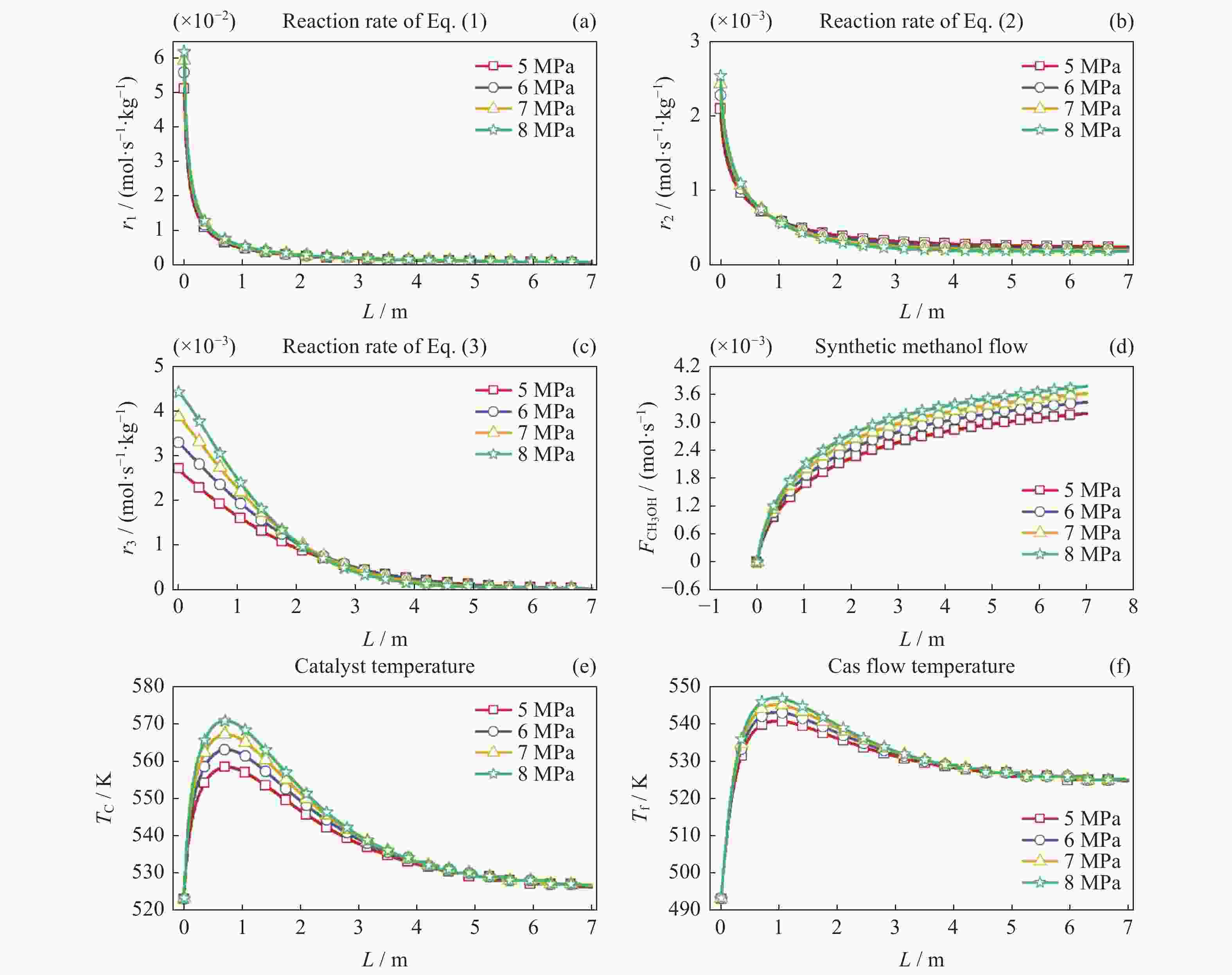
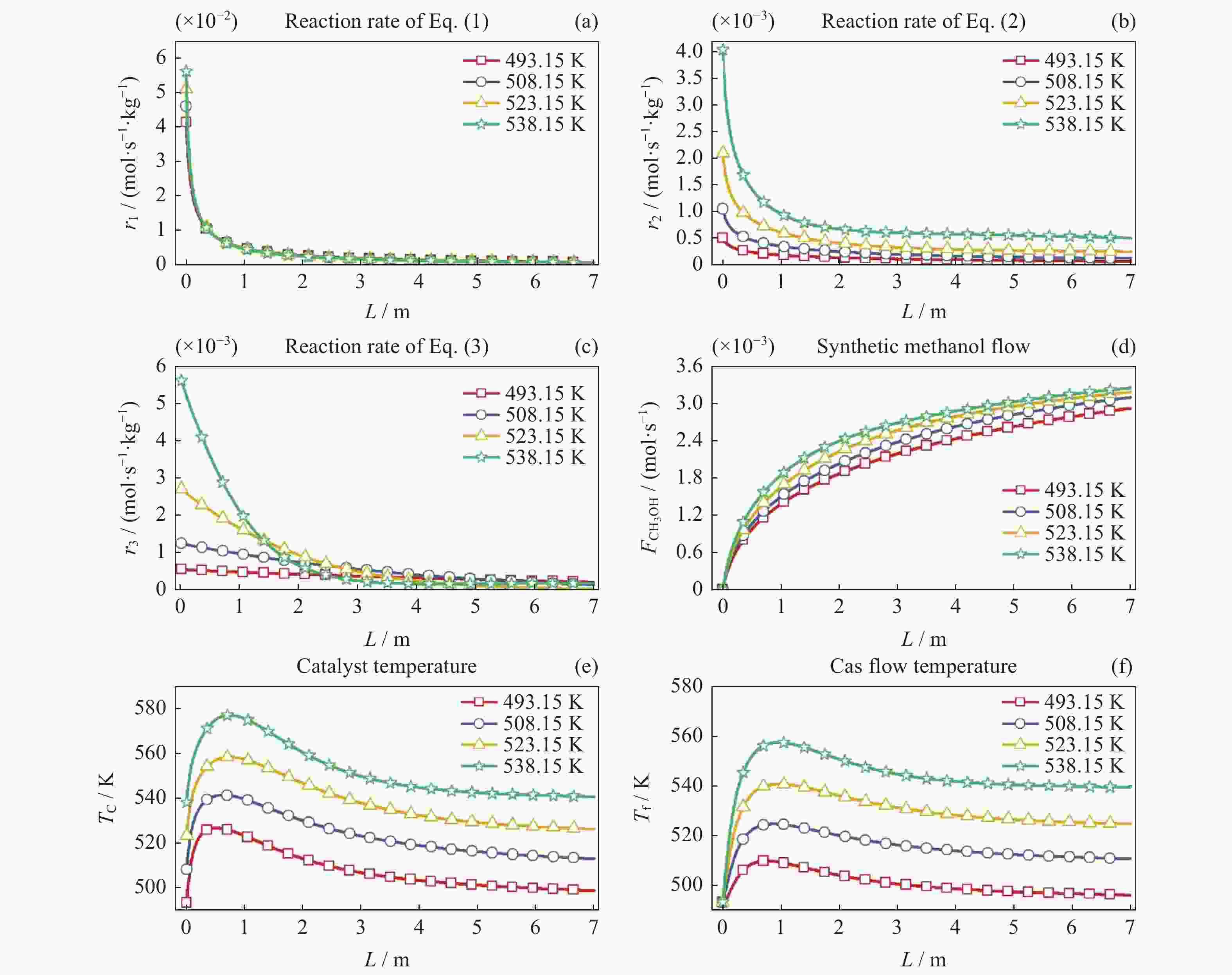
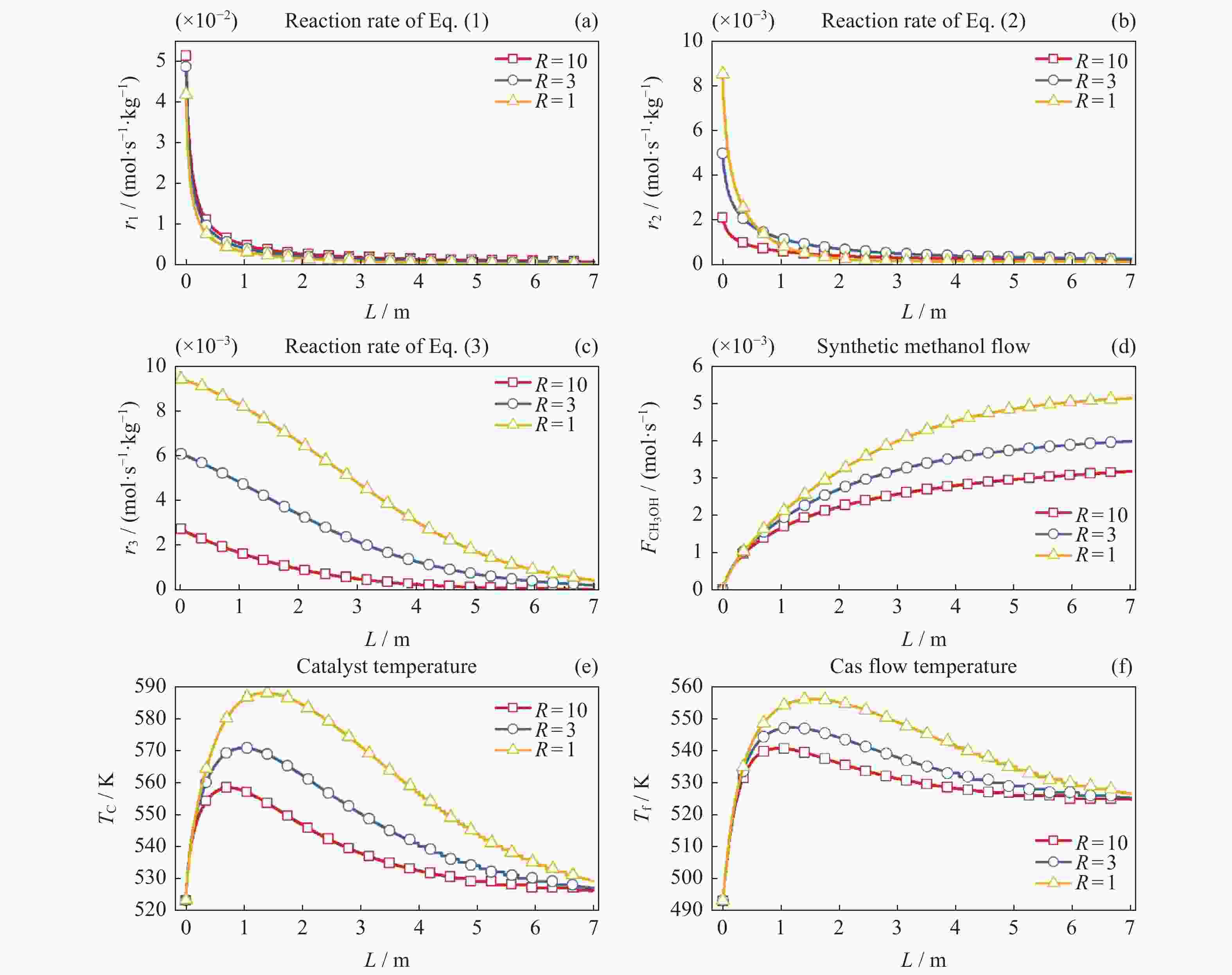
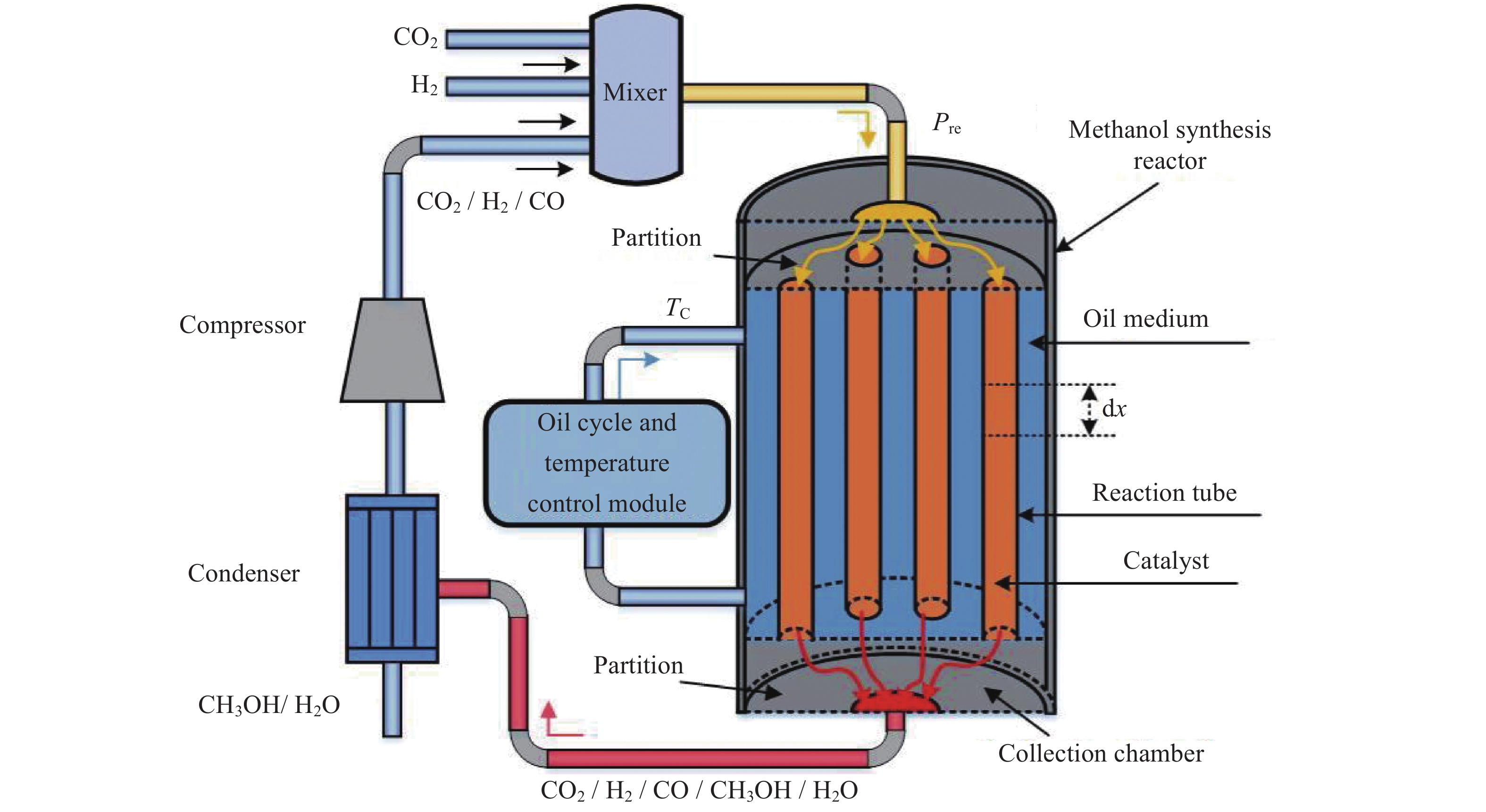
摘要: 在载人密闭空间内通过电解水方式为乘员供氧会产生副产物氢气(H2)。此外,乘员还呼出二氧化碳(CO2)。将H2和CO2催化合成甲醇(CH3OH)是消除载人密闭空间内富余H2和CO2的最优方式之一。对其开展反应过程建模及反应特性研究有助于进行反应过程的控制,更好地维持载人密闭空间内的大气平衡。本文采用微元法建立了H2和CO2催化合成甲醇的物料计算模型和温度一维非均相模型,研究了不同反应压力、冷却介质温度以及入口反应气体中CO2与CO比值等反应条件下的反应特性变化规律。结果显示,反应压力的增加、冷却介质的温升以及入口气体中CO2与CO比值的减小均能促进各反应速率增加,进而使得H2和CO2消除量增加、甲醇合成率上升以及催化剂和反应气体最高温度上升。在保证反应速率增加且催化剂最高温度不超过合理反应温度区间的最大值573.15 K时需维持反应压力不大于8 MPa,冷却介质温度不高于538.15 K以及CO2与CO比值不小于1。Abstract: In the habitable confined space, hydrogen (H2), the by-product of water electrolysis to provide breathing oxygen to crews, and crew’s exhaled carbon dioxide (CO2) are the two surplus gases which should be eliminated for maintenance of atmospheric balance and protection of crew’s life safety. Catalytic synthesis of methanol (CH3OH) from H2 and CO2 is one of the best ways to eliminate surplus H2 and CO2 in manned confined space. The conclusions drawn through reaction modeling and characteristic analysis can guide the control of the reaction process, which is conducive to maintaining the atmospheric balance in confined space. In this paper, based on the established material calculation model and temperature one-dimensional heterogeneous model, the reaction characteristics of catalytic synthesis of methanol from H2 and CO2 under different reaction pressures, coolant temperatures, and CO2 to CO ratios were studied. Results show that the increase of reaction pressure, the growth of coolant temperature, and the decrease of CO2 to CO ratio in inlet gas can all promote the increases of reaction rate, methanol synthesis rate, and the maximum temperatures of catalyst and reactant. Besides, in order to keep the catalyst maximum temperature within a reasonable reaction range (lower than 573.15 K), the reaction pressure should not be higher than 8 MPa, the coolant temperature is not higher than 538.15 K, and the ratio of CO2 to CO is not less than 1.
-
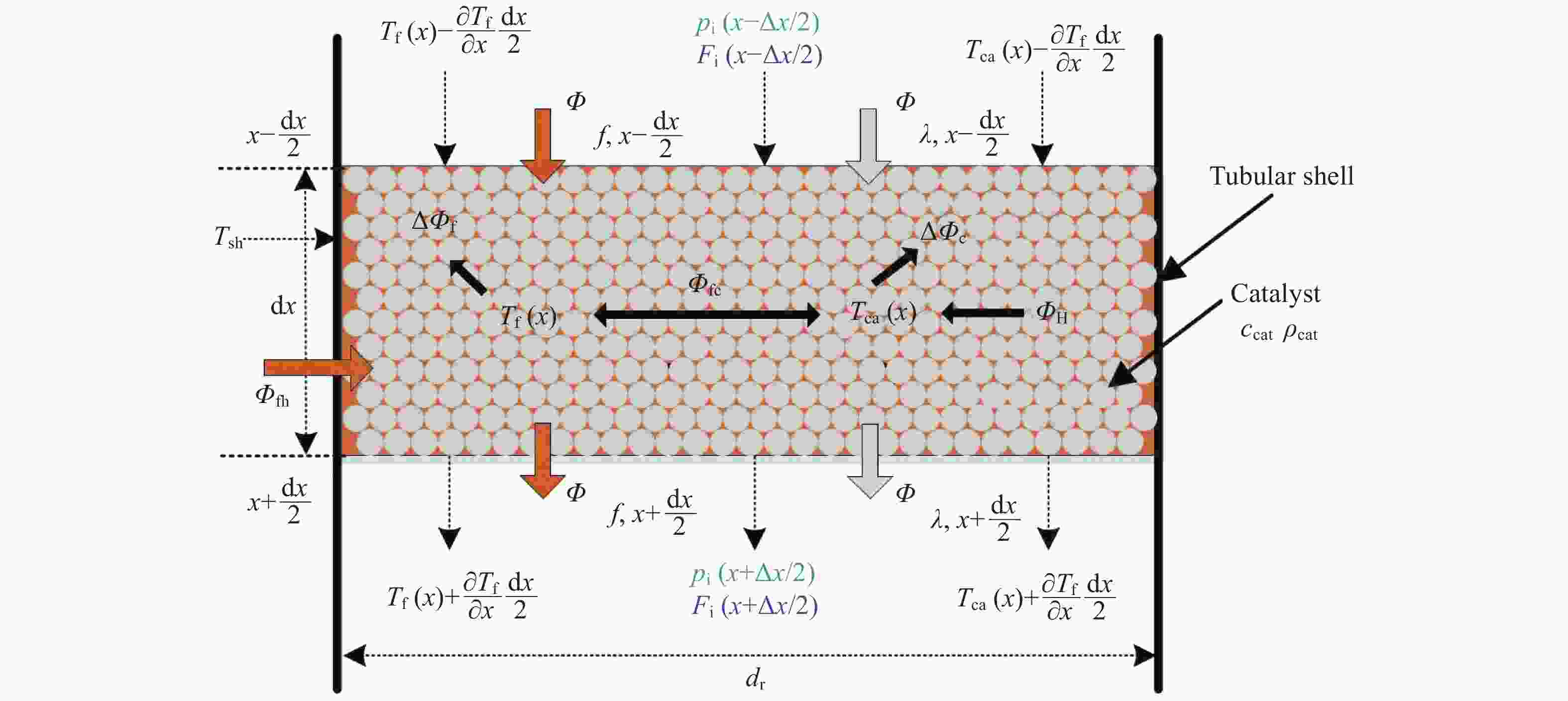
表 1 各个物料成分的反应速率
Table 1. Reaction rate of each material component
${r'_{{{\text{H}}_{\text{2}}}}}$ ${r'_{{\text{C}}{{\text{O}}_{\text{2}}}}}$ ${r'_{{\text{CO}}}}$ ${r'_{{\text{C}}{{\text{H}}_{\text{3}}}{\text{OH}}}}$ ${r'_{{{\text{H}}_{\text{2}}}{\text{O}}}}$ $-3{r_1}\left( x \right)-{r_2}\left( x \right)-{r_3}\left( x \right)$ $ - {r_1}\left( x \right) - {r_2}\left( x \right)$ ${r_2}\left( x \right)-{r_3}\left( x \right)$ ${r_1}\left( x \right){\text{ + }}{r_3}\left( x \right)$ ${r_2}\left( x \right){\text{ + }}{r_3}\left( x \right)$ 表 2 H2和CO2合成甲醇反应工作参数
Table 2. Working parameter sheet of carbon dioxide hydrogenation to methanol
Parameter Value Reaction tube diameter $ {d_{\text{t}}}/{\rm{m}} $ 0.011 Reaction tube length $ L /{\rm{m}}$ 7 Porosity $ \varepsilon $ 0.3 Catalyst density $ {\rho _{\text{c}}}/({\rm{kg}} \cdot {\rm{m}}^{-3}) $ 1770 Inlet gas temperature/K 493.15 Space velocity/h–1 3600 表 3 H2和CO2合成甲醇反应仿真计算工况设计
Table 3. Working condition arrangement of carbon dioxide hydrogenation to methanol
No. Reaction pressure/MPa Coolant temperature/K R(CO2/CO) 1 5,6,7,8 523.15 10 2 5 493.15,508.15,523.15,538.15 10 3 5 523.15 10,3,1 -
[1] 汤兰祥, 高峰, 邓一兵, 等. 中国载人航天器环境控制与生命保障技术研究[J]. 航天医学与医学工程, 2008, 21(3): 167-174TANG Lanxiang, GAO Feng, DENG Yibing, et al. Research on environmental control and life support system (ECLSS) of China’s manned spacecraft[J]. Space Medicine & Medical Engineering, 2008, 21(3): 167-174 [2] ERICKSON R J, HOWE J, KULP G W, et al. International space station united states orbital segment oxygen generation system on-orbit operational experience[J]. SAE International Journal of Aerospace, 2008, 1(1): 15-24 [3] CICHAN T, BAILEY S A, ANTONELLI T, et al. Mars base camp: an architecture for sending humans to mars[J]. New Space, 2017, 5(4): 203-218 [4] 李俊荣, 尹永利, 周抗寒, 等. 空间站电解制氧技术研究进展[J]. 航天医学与医学工程, 2013, 26(3): 215-220LI Junrong, YIN Yongli, ZHOU Kanghan, et al. Progress of oxygen generation technology by water electrolysis in space station[J]. Space Medicine & Medical Engineering, 2013, 26(3): 215-220 [5] 郑东欢, 李玉文, 陈兆波, 等. 水下航行器舱室内CO2的去除及O2再生工艺系统设计[J]. 环境工程学报, 2009, 3(1): 137-142ZHENG Donghuan, LI Yuwen, CHEN Zhaobo, et al. Technical system design of exhaust gas disposal and oxygen regeneration in cabin of aircraft ocean-below[J]. Chinese Journal of Environmental Engineering, 2009, 3(1): 137-142 [6] 李军, 艾尚坤, 周抗寒. 空间站Sabatier CO2还原装置实验研究[J]. 航天医学与医学工程, 1999, 12(2): 121-124LI Jun, AI Shangkun, ZHOU Kanghan. An experimental study of the Sabatier CO2 reduction subsystem for space station[J]. Space Medicine & Medical Engineering, 1999, 12(2): 121-124 [7] LEONZIO G, ZONDERVAN E, FOSCOLO P U. Methanol production by CO2 hydrogenation: analysis and simulation of reactor performance[J]. International Journal of Hydrogen Energy, 2019, 44(16): 7915-7933 [8] WANG G, MAO D S, GUO X M, et al. Enhanced performance of the CuO-ZnO-ZrO2 catalyst for CO2 hydrogenation to methanol by WO3 modification[J]. Applied Surface Science, 2018, 456: 403-409 [9] XIAO J, MAO D S, WANG G, et al. CO2 hydrogenation to methanol over CuO-ZnO-TiO2-ZrO2 catalyst prepared by a facile solid-state route: the significant influence of assistant complexing agents[J]. International Journal of Hydrogen Energy, 2019, 44(29): 14831-14841 [10] SEIDEL C, JÖRKE A, VOLLBRECHT B, et al. Kinetic modeling of methanol synthesis from renewable resources[J]. Chemical Engineering Science, 2018, 175: 130-138 [11] GRAAF G H, STAMHUIS E J, BEENACKERS A A C M. Kinetics of low-pressure methanol synthesis[J]. Chemical Engineering Science, 1988, 43(12): 3185-3195 [12] MANENTI F, CIERI S, RESTELLI M. Considerations on the steady-state modeling of methanol synthesis fixed-bed reactor[J]. Chemical Engineering Science, 2011, 66(2): 152-162 [13] SHAHROKHI M, BAGHMISHEH G R. Modeling, simulation and control of a methanol synthesis fixed-bed reactor[J]. Chemical Engineering Science, 2005, 60(15): 4275-4286 [14] KISS A A, PRAGT J J, VOS H J, et al. Novel efficient process for methanol synthesis by CO2 hydrogenation[J]. Chemical Engineering Journal, 2016, 284: 260-269 [15] DOS SANTOS R O, DE SOUSASANTOS L, PRATA D M. Simulation and optimization of a methanol synthesis process from different biogas sources[J]. Journal of Cleaner Production, 2018, 186: 821-830 [16] BATHE K J, WILSON E L. Stability and accuracy analysis of direct integration methods[J]. Earthquake Engineering & Structural Dynamics, 1972, 1(3): 283-291 -
-






 下载:
下载: